A First-in-Class ANTI SOLUBLE FAS LIGAND
for Severe immune-mediated skin blistering diseases
Transforming care for
Severe immune-mediated skin blistering diseases
Award Winning Approach
PC111 addresses the urgent unmet need in conditions like Pemphigus Vulgaris and SJS/TEN by selectively blocking the FasL-driven apoptotic pathway. This groundbreaking, non-immunosuppressive therapy offers a novel, disease-modifying solution with strong therapeutic and commercial potential. PC-111 is an in-licensed asset from PinCell Srl, an academic spin-off of the University of Modena-Reggio Emilia, Italy.
The approach behind PC111 was recognized with the “Oscar of Italian Dermatology” for its award-winning scientific innovation.

The Unmet Need
A Critical Gap in Targeted Therapies for
Severe Blistering Skin Diseases
Severe rare blistering skin disorders remain critically underserved, with high morbidity, mortality, and no effective targeted treatments. Current therapies fail to address the fundamental biological mechanisms-keratinocyte apoptosis and blister formation-highlighting the urgent need for safe, mechanism-based approaches.
Pemphigus Vulgaris (PV)
A rare autoimmune blistering disease currently treated with non-specific immunosuppression, often resulting in relapse, increased infection risk, and significant toxicity.

~300,000
patients worldwide
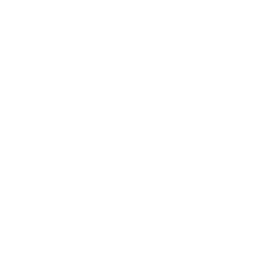
5–15%
mortality
despite current care
Stevens-Johnson Syndrome / Toxic Epidermal Necrolysis (SJS/TEN)
A severe, life-threatening skin disorder with no approved therapies and limited supportive care options.

5,000–10,000
patients worldwide

up to 30%
mortality
rates

A Blockbuster Drug Opportunity
Both indications represent a >$1B commercial opportunity and address a massive unmet need in rare skin disease treatment.
THE GAP IN TODAY’S TREATMENT LANDSCAPE
Existing treatments suppress the immune system but do not directly target disease pathology.
Key limitations
Broad immunosuppression with significant safety burden
Delayed and incomplete responses
Frequent relapse
Poor long-term tolerability
Critically, the FasL-driven apoptotic pathway remains unaddressed.
Our Solution
PC111
PC111 blocks the blister formation and restores skin integrity
How it works?
Differentiated Mechanism of Action
By intervening upstream in disease biology, PC111 addresses pemphigus and related disorders at their source.
Neutralizes
pathogenic Soluble
Fas Ligand sFasL
Preserves
membrane-bound
FasL (mFasL)
essential for immune
homeostasis
Enables a non-immunosuppressive therapeutic approach
first-in-class, fully human monoclonal antibody.
Selectively targeting soluble Fas Ligand (sFasL)-a key mediator of blister formation.
Treatment Benefits

First-in-Class
Anti-sFasL Therapy
Targets a previously unaddressed disease-driving pathway.

Non-Immunosuppressive Mechanism
Preserves immune homeostasis while controlling disease.

High Selectivity & Safety Potential
Specific for sFasL with reduced risk of off-target effects.

Broad Disease Applicability
Potential to treat multiple severe skin disorders, including pemphigus and SJS/TEN.

Pre-Clinical Results
PC111 has demonstrated robust efficacy across translational models.
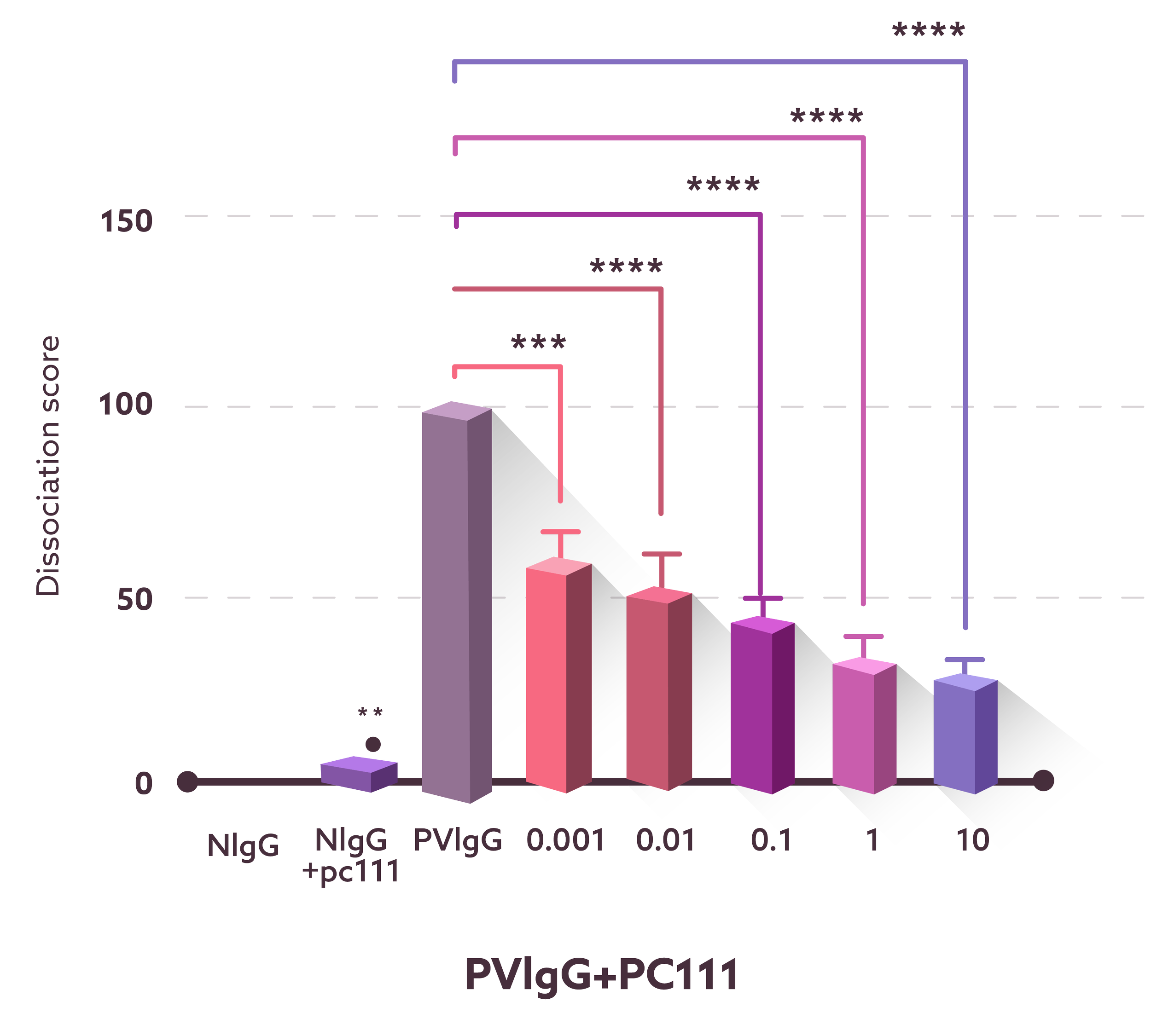
In vitro
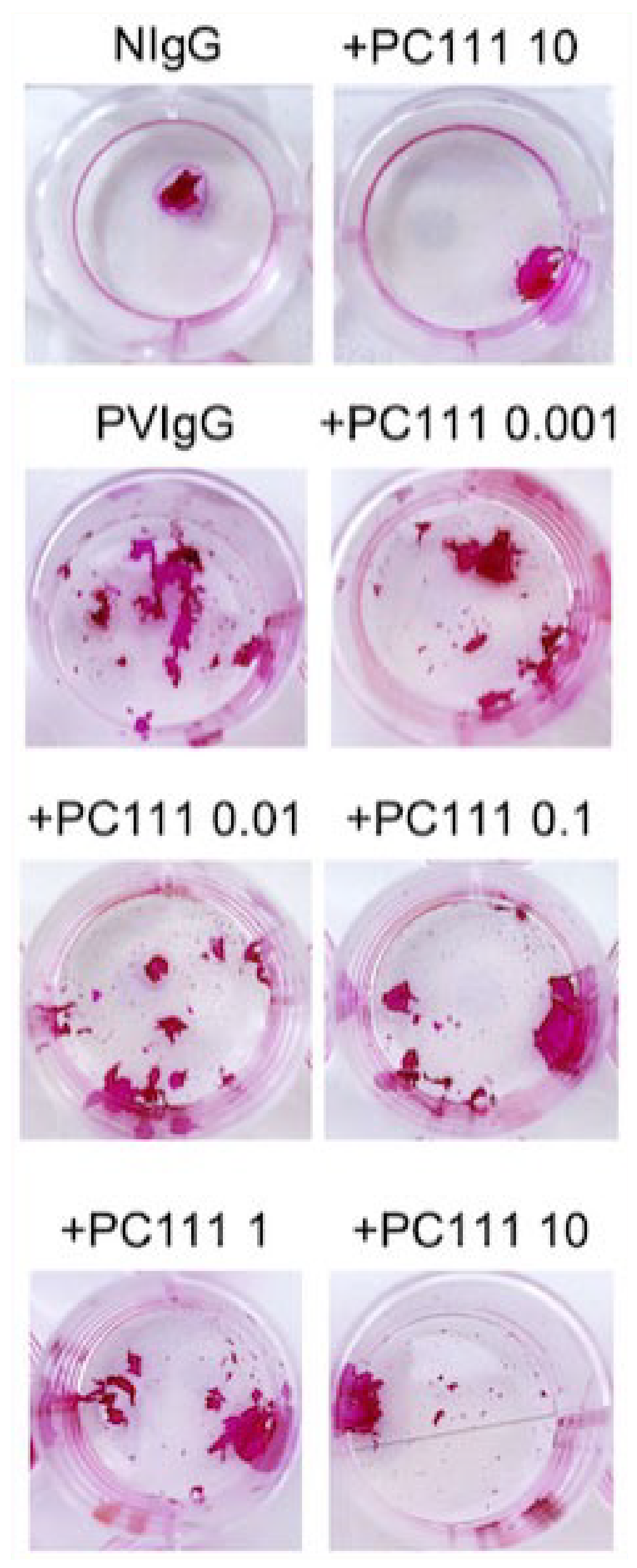
PC111 preserves keratinocyte cell–cell adhesion in an in vitro pemphigus model.
Keratinocytes were incubated with normal human IgG (NIgG) or pemphigus vulgaris IgG (PVIgG), with or without increasing concentrations of PC111 (0.001–10 µg/mL). Cell–cell adhesion was assessed using a dispase-based dissociation assay. Purple areas indicate intact keratinocyte cell sheets or fragments. Representative images show that PVIgG induced marked cell sheet fragmentation (acantholysis), while PC111 dose-dependently reduced fragmentation and preserved epithelial integrity.
Quantification was performed by counting cell sheet fragments and expressed as a dissociation score, as shown in the bar graph.
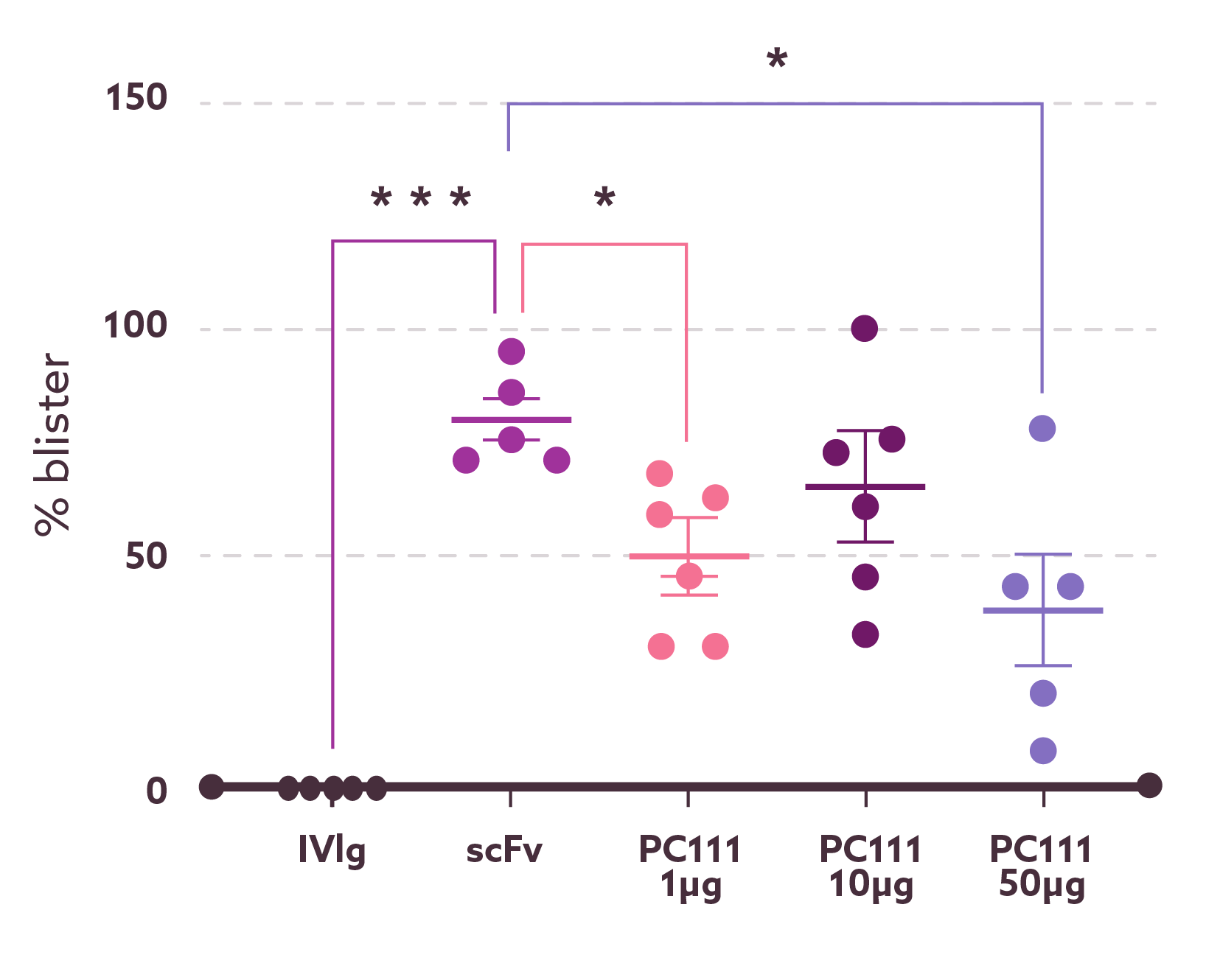
Ex vivo
(Pemphigus models)
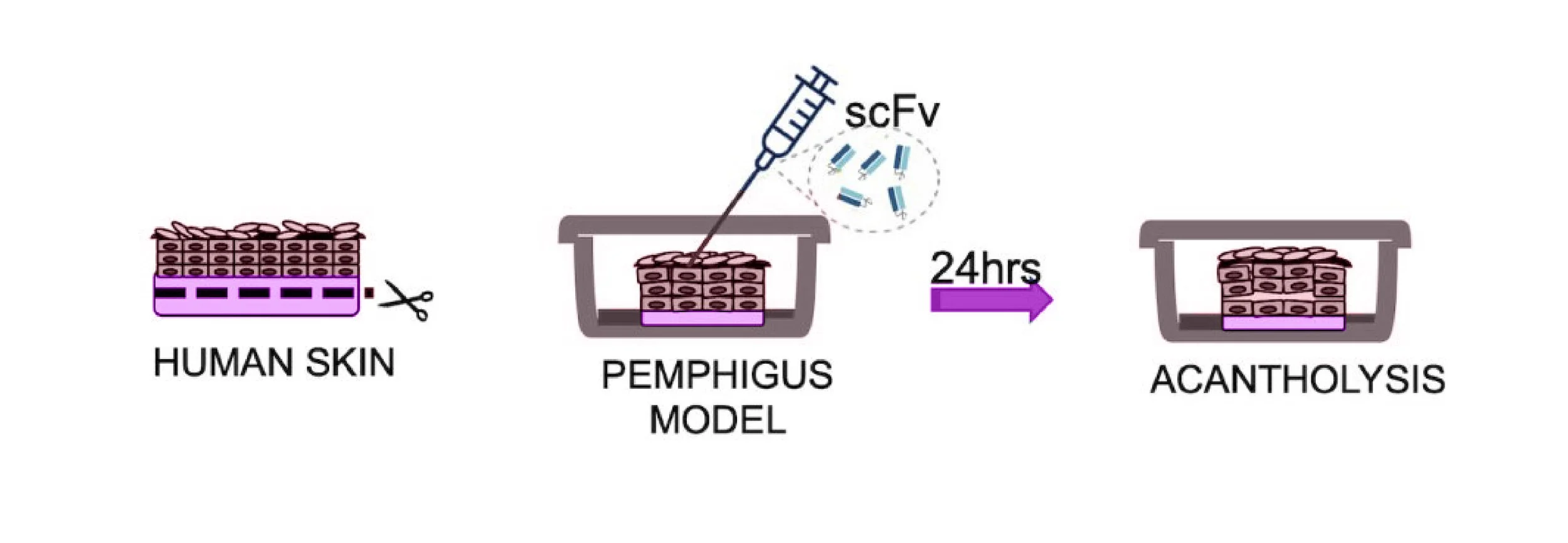
PC111 inhibits blister formation in an scFv-induced ex vivo pemphigus model.
A schematic illustration shows the experimental setup in which human skin samples were exposed to pemphigus-inducing scFv, resulting in acantholysis and blister formation within 24 hours. Blister formation was quantified by measuring blister length, expressed as a percentage of the total skin length analyzed. PC111 treatment significantly reduced blister formation in a dose-dependent manner with up to a 50% reduction.
In vivo
Efficacy confirmed in the first humanized FasL mouse model
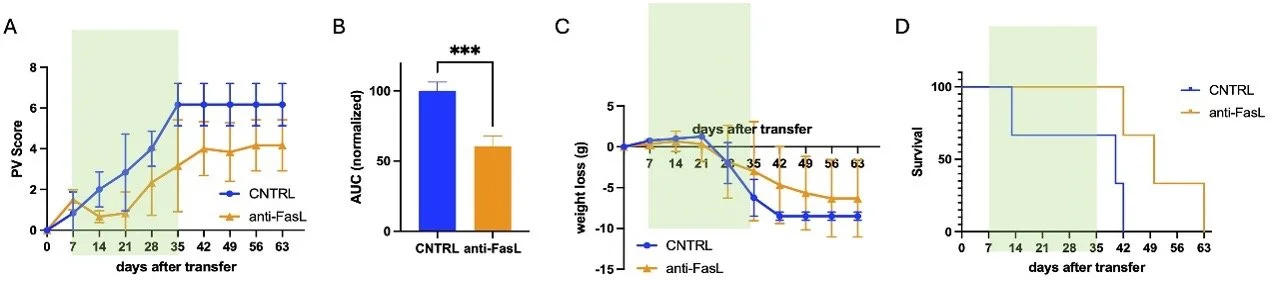
Effects of anti-sFasL antibody (PC111) treatment in an active pemphigus mouse model (DSG1/DSG3 autoreactive).
Disease progression and treatment response were evaluated over time. (A) Clinical severity was assessed using the PV score. (B) Overall disease burden was summarized as area under the curve (AUC) of the PV score over time. (C) Body weight changes were monitored as an indicator of disease burden. (D) Survival was recorded throughout the study period. Anti-sFasL treatment significantly reduced disease severity, limited weight loss, and improved survival compared with control animals.
Validated Efficacy
Future Plans & Timeline
Program: PC111
Indication: Pemphigus, SJS/TEN
Licensor: Pincell SRL
Patent: PCT issued in 2023
Ex-vivo PoC- proving MoA in skin tissue
Done
First in-vivo PoC – efficacy in live psoriatic tissue
Done
Second in-vivo PoC –
duration of effect
H1 2026
Toxicology
H2 20 26

Contact us
Get in touch to explore partnership opportunities and consult our CDMO specialists




