Scinai Bioservices
Biologics CDMO
End-to-End Drug Development & Manufacturing
Scinai CDMO Bioservices
Comprehensive Biologics CDMO Services
From early research to Phase 3 clinical trials
Scinai BioServices supports biopharma innovators from early research to late-phase clinical production, delivering agile, cost-effective, and science-driven CDMO solutions backed by over 20 years of experience and a state-of-the-art cGMP facility.
Our holistic, partnership-focused approach ensures process excellence, accelerated timelines, and value beyond the project, helping innovators bring their therapies to life with confidence.
Scinai CDMO Bioservices
Comprehensive Biologics CDMO Services
From early research to Phase 3 clinical trials

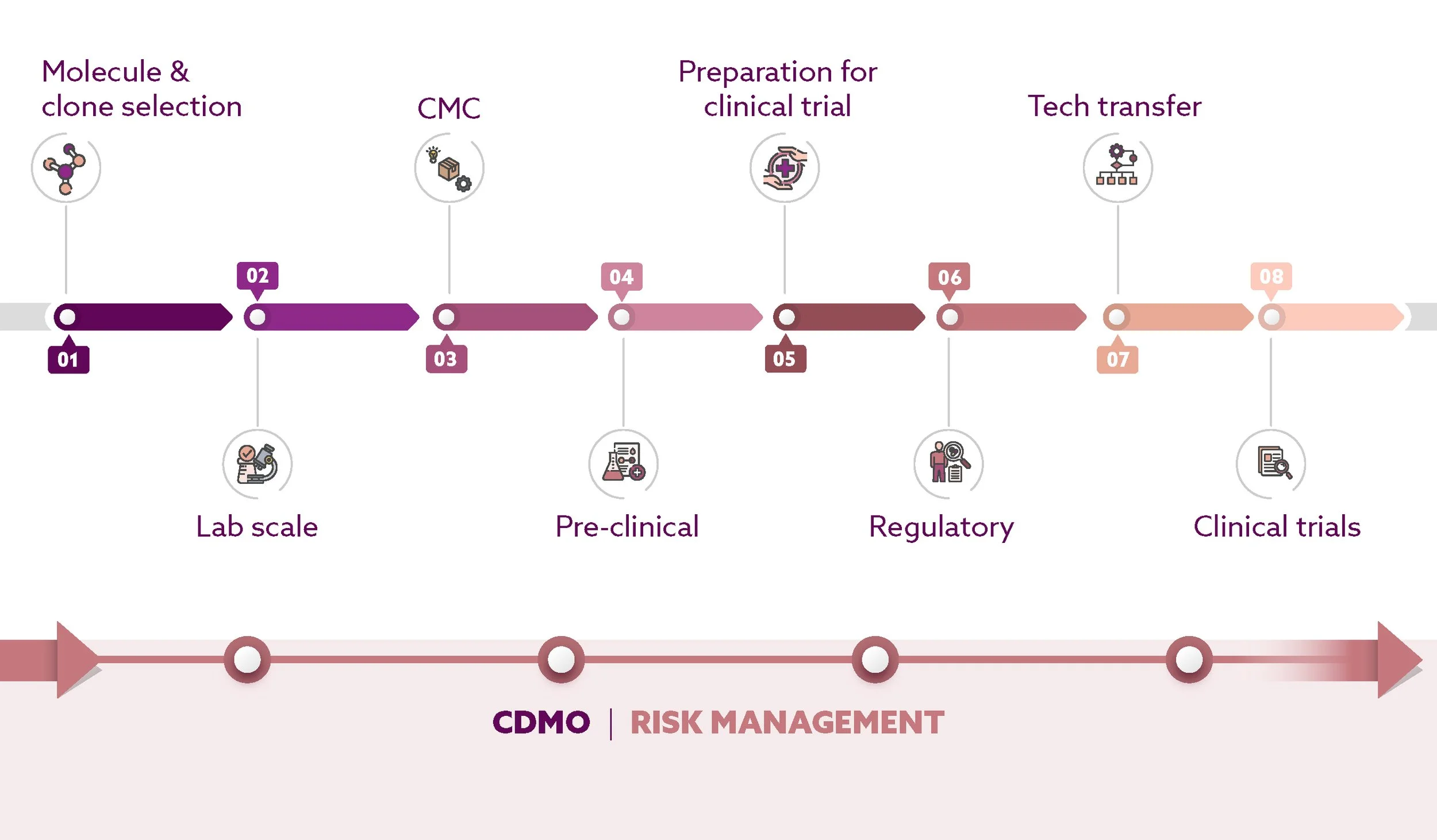
MOLECULE & CLONE SELECTION
LAB SCALE

CMC

PRE-CLINICAL

PREPARATION FOR CLINICAL TRIAL

REGULATORY

TECH TRANSFER

CLINICAL TRIALS


Scinai BioServices supports biopharma innovators from early research to late-phase clinical production, delivering agile, cost-effective, and science-driven CDMO solutions backed by over 20 years of experience and a state-of-the-art cGMP facility.
Our holistic, partnership-focused approach ensures process excellence, accelerated timelines, and value beyond the project, helping innovators bring their therapies to life with confidence.



1,850m² Biologics cGMP Manufacturing facility
State of the art Development and Manufacturing facility 15M+ euro invested in cutting edge equipment
High-end manufacturing suites designed to meet FDA and EMA cGMP standards and have successfully passed European QP (EMA) and Israeli MoH audits for Phase 3 clinical trial production.

State of the art laboratories
Equipped with cutting-edge analytical and bioprocessing technologies, including ÄKTA systems for purification, Maurice for protein characterization, Octet for real-time binding analysis and the AMBR250 fermenter for automated, high-throughput cell culture and process optimization.

STERILE Processing
cGMP aseptic fill and finish
Our aseptic processing capabilities enable the fill and finish of vials and pre-filled syringes (PFS) for clinical trials, ensuring full compliance with FDA and EMA cGMP standards. The automated filling machine, operating under Grade A RABS within a Grade B cleanroom, ensures maximum sterility, precision, and consistency across various container types and volumes - supporting reliable scale-up from clinical to commercial manufacturing.

Protein expression Solutions - Expertise in bacterial and yeast systems:
Process Development & Scale-Up
With extensive expertise in recombinant protein development, Scinai delivers cost-effective, flexible, and scalable process development and scale-up solutions. Our experts guide clients through each stage of research and development, helping to minimize risks and accelerate progress toward clinical success.

Comprehensive Protein Analytical Solutions:
Analytical Methods Development
Our experienced team of scientists utilizes advanced analytical technologies to ensure precise and reliable product characterization. We provide comprehensive analytical method development for in-process control, release testing, and detailed analysis of recombinant proteins.
Scinai Bioservices Inc.
U.S Subsidiary
Our Delaware based U.S. subsidiary, provides access to federal funding under the Bio Secure Act.
Top-tier pharma expertise
20 + years of experience
Trusted by…
EMPOWERING BIOTECH INNOVATORS.
WHAT DO OUR CUSTOMERS SAY

Scinai Immunotherapeutics is proud to serve as the CDMO partner for Hopec Pharma, supporting the GMP manufacturing of DOR-100, an innovative therapy for Non-Muscle Invasive Bladder Cancer (NMIBC).
Hear from Hopec Pharma’s Co-Founders as they share insights about the project and the unique value Scinai brought to its development.


Serpin Pharma partnered with Scinai BioServices for their Phase 2 clinical trial in Germany and Israel, marking the first use of Scinai’s boutique CDMO services.
The project involved bulk formulation, aseptic filling, stability studies, the development of analytical release methods, and compliance with international regulatory standards.
-
Dr. Cohava Gelber, CEO of Serpin Pharma, shared:
"We appreciated Scinai’s dedication, customized approach, and high level of professionalism and expertise throughout the project. The team operated with modern, well-equipped labs and demonstrated strong capabilities to scale and adapt to our evolving needs. Most importantly, they were attentive, reliable, and fully aligned with the stringent regulatory standards required for our clinical program."

״We chose Scinai as our partner because they provide a genuine end-to-end CDMO solution and deliver value that exceeds a standard project. Their expertise, speed, and flexibility are precisely what a startup like ours requires.״
Dan Touitou, CEO

״We selected Scinai due to their vast experience in bio-pharmaceutical drug development and manufacturing for clinical trials while meeting stringent international regulatory requirements. The pristine condition of Scinai’s modern and highly equipped laboratories and clean rooms demonstrate the competence and attention to detail that are imperative for a CDMO to successfully serve our future needs.״
Yanir Aldouby, COO

״Scinai provided us with professional advice and support at the start of our journey. We received timely professional responses, with thoughtful attention to our requests.״
Nir Netzer, COO

״We were looking for a professional partner for the ongoing development and production of our product. Scinai was exactly what we were looking for in a partner. We found their team to be professional, reliable, and particularly attentive to our needs, demonstrating the flexibility we needed.״
Ohad Lavi, CEO









cdmo FAQ
-
We provide end-to-end CDMO solutions for biologics, supporting companies from early-stage research through late-phase clinical trials. Our services include: sterile processes and aseptic filling, injectables vials and syringes, analytical method development, upstream and downstream process development and scale-up, bulk formulation, stability studies, regulatory documentation, and cGMP manufacturing.
-
Our state-of-the-art laboratories are in Jerusalem, Israel, with our U.S. subsidiary Scinai Bioservices Inc. to support North American clients. This enables seamless collaboration across time zones and regulatory markets.
-
We specialize in recombinant proteins, bacteria and yeast, peptides, formulation of lipid and liposomes (LNP), and other advanced therapeutics, providing tailored solutions to meet the unique requirements of each molecule and clinical stage.
-
As a boutique CDMO, we offer flexibility, agility, and highly personalized service. Our teams provide hands-on support, adapting to your project’s evolving needs while maintaining scientific rigor and speed to clinic.
-
Yes. Scinai offers full sterile processing capabilities, including not only aseptic fill & finish but also the preceding sterile preparation and handling steps.
These sterile operations are mandatory for certain drug categories, particularly large biomolecules, insoluble or poorly soluble proteins, large liposomes and other lipid-based nanoparticles, microbial therapeutics, including both Gram-negative and Gram-positive organisms. -
The Scinai facility is purpose-built for sterile manufacturing, featuring dedicated suites, controlled personnel and material flows, and a hybrid single use and closed-system equipment.
The Scinai team brings extensive hands-on experience, having successfully executed multiple manufacturing campaigns that involve critical sterile preparations.
-
Yes. Scinai’s GMP facility is designed and operated to meet both FDA and EMA current Good Manufacturing Practice (cGMP) standards. This includes cleanrooms, aseptic processing suites, sterile fill & finish, and supporting quality systems built according to U.S. and European regulatory expectations.
The facility has recently successfully passed European QP (EMA) and Israeli Ministry of Health audits -demonstrating compliance with stringent GMP requirements.
Furthermore, Israel has an international agreement under which its pharmaceutical manufacturing standards (especially Good Manufacturing Practice, GMP) are recognized as equivalent to European standards. As a result, Israel and EU authorities mutually accept each other’s GMP inspection conclusions

Contact us
Get in touch to explore partnership opportunities and consult our CDMO specialists







